Decaf coffee has changed significantly in recent years. Advances in processing methods and roasting practices…
Decaffeinated coffee can be produced using several different methods, from solvent extraction to water filtration and supercritical CO₂ processing. Each approach removes caffeine in its own way and can subtly influence how the coffee tastes in the cup. In this guide, we explain how modern decaffeination works, compare the main methods, and look at what James Hoffmann’s Decaf Project revealed about their real impact on flavor.
This article is part of CoffeeHow’s research series on decaf coffee, based on industry sources, scientific publications, and practical tasting comparisons.
If you’re new to decaf coffee or want a full overview, start with our main guide: Decaf Coffee: What It Is, How It’s Made, and How to Choose.
Before scientists could understand how to decaffeinate coffee, they first had to learn how to isolate caffeine as a compound. However, nobody initially set out to decaffeinate coffee. The experiments that led to the isolation of caffeine in coffee were originally used to compare it with belladonna extract.
Decaf coffee didn’t start as a planned invention. It came about by accident. In 1903, German coffee merchant Ludwig Roselius noticed that a shipment of coffee beans had been soaked with seawater during transport. Most of the caffeine was washed out, while the coffee’s flavor remained largely intact. This observation helped lay the groundwork for the first commercial decaffeination process.
The earliest commercially viable decaffeination methods relied on chemical solvents, and they were not exactly gentle. One of the first widely used approaches was developed by German coffee merchant Ludwig Roselius in 1905. His process removed caffeine from pre-moistened green coffee beans using benzene, a solvent that is now recognized as toxic. That is one of the reasons this early technique is widely considered outdated and controversial today.

Benzene is no longer used for decaffeination. Modern solvent-based decaf typically relies on ethyl acetate (EA) or methylene chloride instead. The U.S. FDA allows ethyl acetate to be used as a solvent for decaffeinating coffee under current good manufacturing practice, and it also limits the trace residue of methylene chloride in decaf coffee to no more than 10 parts per million (ppm). In other words, when used correctly, the remaining amounts are extremely small, and most of the safety concerns relate to the chemical at higher exposures rather than the tiny residue levels found in brewed decaf coffee.
Ethyl acetate (EA) can be naturally derived, and it’s widely considered a safe food-grade solvent. It also occurs naturally in many fruits and vegetables, which is why you’ll often see it described as a “naturally occurring” compound.
With methylene chloride, the key point is dosage: modern decaf processing leaves only trace amounts, and those traces are tightly regulated. The FDA limits methylene chloride residue in decaffeinated coffee to no more than 10 ppm, which is an extremely small amount.
To put that into perspective:
10 ppm = 10 mg per 1 kg of coffee.
In terms of brewed coffee, 1 kg of beans is roughly enough for about 66 pour-overs (15 g each)
or about 55 double espressos (18 g each).
(These cup numbers are calculated from the FDA limit and typical brew doses.)
Now, coffee can be decaffeinated in a few different ways.
Most methods still use a solvent-based decaffeination process, though with food-grade, human-safe solvents.

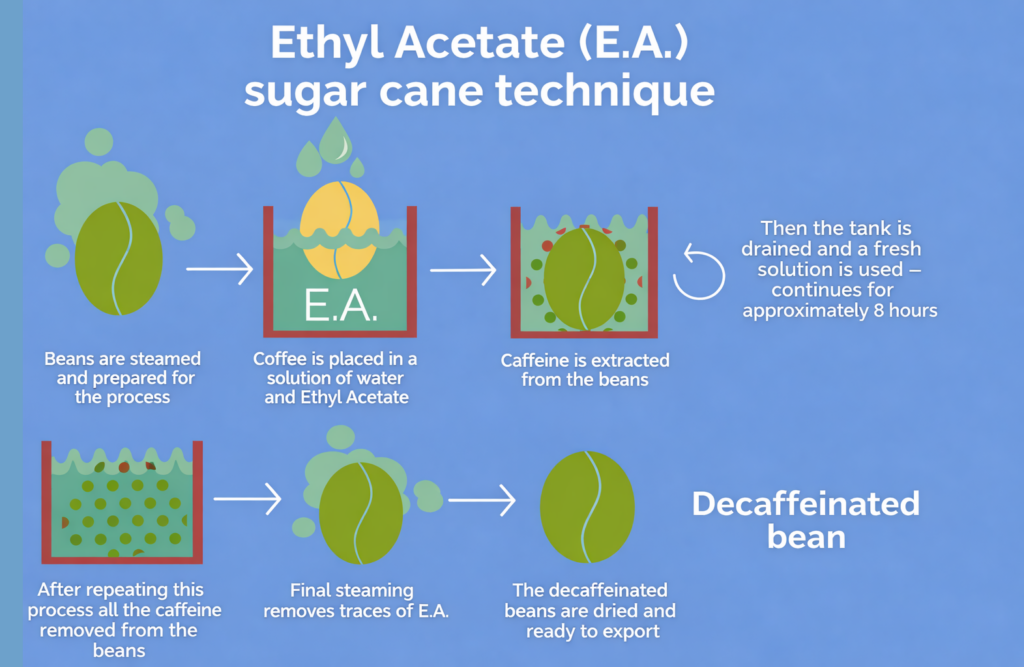
Depending on how it is presented, this method may sound either technical or surprisingly appealing. Some roasters call it the ethyl acetate (EA) process, while others market it as the sugarcane process. The name matters because ethyl acetate can be naturally derived. It occurs naturally in fruits and vegetables, and it can also be derived from molasses, a byproduct of sugar production. This is where the “sugarcane” label comes from.
However, ethyl acetate can also be synthetically produced, so if a fully “natural” EA process matters to you, it’s worth checking with your roaster where and how the decaffeination is done.
As for the process itself, it’s a classic solvent-style decaffeination method. Green coffee beans are first gently steamed to open up their structure and prepare them for extraction. Then they’re washed with ethyl acetate, which binds with the caffeine in the bean and extracts it while leaving most of the other flavor compounds. After the desired caffeine level is reached, the EA residue on the beans is removed by steaming them.
This method is popular for a reason. It typically removes around 97% or more of caffeine while keeping the coffee’s flavor profile largely intact, making it one of the most practical ways to produce great-tasting decaf at scale.

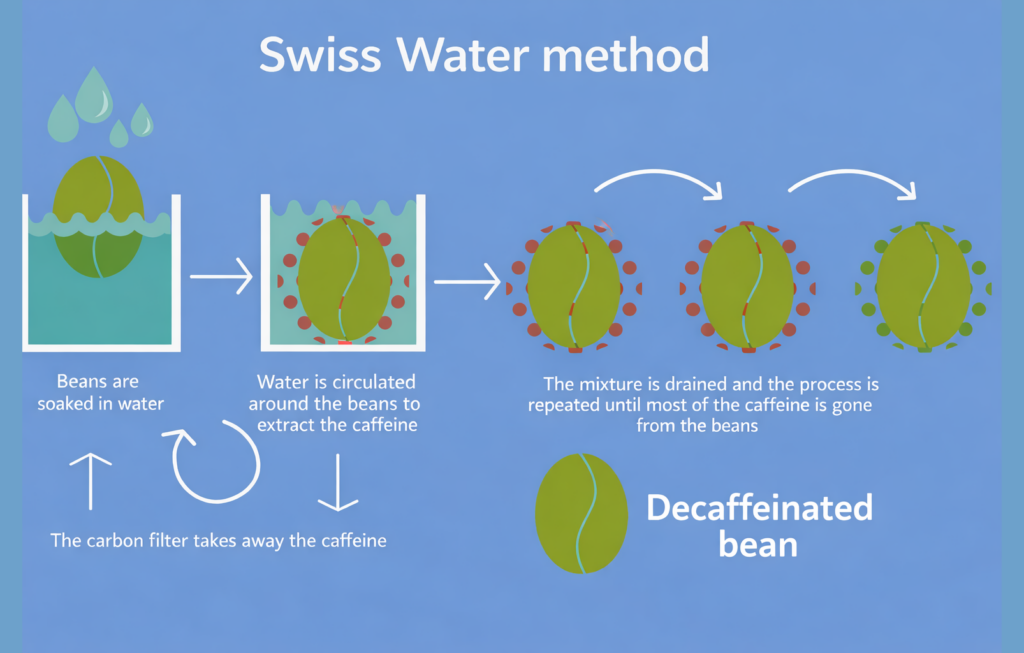
The Swiss Water Process is one of the most popular solvent-free ways to make decaf. It’s usually more expensive than solvent methods, but it’s widely valued for its clean approach because it uses only hot water, carbon filtration, and time.
Because no chemical solvents are added, some coffee drinkers describe Swiss Water decaf as tasting “cleaner” or less “processed,” although taste is always subjective. One funny detail is that despite the name, Swiss Water coffee isn’t actually processed in Switzerland. The Swiss Water Decaffeinated Coffee Company is based in British Columbia, Canada (near Vancouver).
Here’s how it works. First, a batch of green coffee is soaked in hot water to create a concentrated green coffee extract (GCE). This pulls out caffeine along with the coffee’s soluble flavor compounds.
Next, the extract is passed through activated carbon filters, which trap the caffeine molecules. What’s left is a caffeine-free extract that still contains most of the coffee’s flavor compounds.
That same extract is then used to soak a fresh batch of green beans. Since the extract is already “full” of flavor compounds, the main thing that moves out of the new beans is caffeine, which helps the coffee keep much of its natural taste. The cycle is repeated until the target caffeine level is reached.
The result is a decaf method that can remove up to about 99.9% of caffeine while preserving much of the coffee’s original character, without using chemical solvents.

The Mountain Water Process is a popular solvent-free way to decaffeinate coffee using water-based extraction. It’s often chosen for a balanced approach that removes caffeine while helping the coffee keep much of its natural character.
One important detail is that “Mountain Water” is not one single universal method. Different producers may use slightly different water-based variations under this label. In many cases, Mountain Water decaf refers to coffee processed in Mexico by DESCAMEX (Descafeinadora de Café de México), using its branded method called MW D’caff®.
Here’s how it works. First, the green coffee is analyzed so the decaffeination parameters can be adjusted for that specific lot. Then the beans are gently steamed and prepared for caffeine extraction.
Next, caffeine is removed using a controlled water-based process. DESCAMEX describes this stage as managed through variables like flow, temperature, pressure, and vacuum, with the goal of removing caffeine while preserving the coffee’s flavor structure.
After extraction, the beans are dried back to a stable moisture level. DESCAMEX highlights a triple drying system as part of its finishing stage, helping stabilize the decaffeinated green coffee before roasting.
The result is a solvent-free decaf method that’s widely used for high-quality Mountain Water coffees from Mexico.
Both Swiss Water and Mountain Water are solvent-free, water-based decaf methods designed to remove caffeine while preserving flavor. Swiss Water is a standardized process run by a single company in Canada, while Mountain Water is a broader label that often refers to Mexican decaf processed by DESCAMEX (MW D’caff®). The key difference is that DESCAMEX emphasizes industrial control parameters like pressure and vacuum, plus a dedicated drying stage, while Swiss Water is best known for its water + carbon filtration cycle.

The CO₂ process is one of the most advanced and less common ways to decaffeinate coffee. It’s expensive to run, which is why it’s not used as widely as water or solvent methods.
This method uses carbon dioxide in a supercritical state, meaning it behaves like both a gas and a liquid at the same time. In this form, CO₂ can move through the coffee bean efficiently while selectively dissolving caffeine under high pressure.
During the process, green coffee beans are placed in a sealed chamber, and pressurized CO₂ is pumped in. The CO₂ selectively extracts caffeine from the beans while aiming to preserve much of the coffee’s original flavor structure.
The caffeine-loaded CO₂ is then moved into a separate stage, where caffeine is removed (typically through filtration), and the CO₂ is recycled and used again.
The result is a decaf method known for strong caffeine removal and minimal flavor impact in many coffees, although the equipment and operating costs are a major reason it’s less common in the specialty market.

Decaffeination methods have been studied for decades in scientific research. These studies typically focus on how efficiently caffeine is removed and how the chemical composition of coffee changes during processing. For example, laboratory work often compares methods like Swiss Water and ethyl acetate by measuring residual caffeine using chromatography and other analytical techniques.
But coffee isn’t just chemistry. Its flavor is shaped by hundreds of compounds that interact and transform during roasting and brewing. Even small compositional changes can be perceived differently in the cup, and sensory perception is always to some degree subjective. Chemistry can tell us how caffeine is removed, but it doesn’t always explain how flavor changes. To understand that, coffee needs to be compared in the cup, which is exactly what James Hoffmann set out to do with The Decaf Project.

A rare real-world comparison of decaffeination methods
For the project, around 16 tons of the same green coffee were sourced from Caravela Coffee and divided into four equal lots.
One portion remained untouched as a caffeinated control sample. The other three were sent for decaffeination using different methods: Supercritical CO₂ processing, Ethyl Acetate Decaffeination, Swiss Water processing
Although decaffeination methods are well understood technically, they are rarely compared in a way that allows differences to be evaluated directly in the cup. That’s what made James Hoffmann’s Decaf Project such an unusual and ambitious experiment.
After processing, all four versions were brought back together, packaged, and distributed to roughly 50 roasters worldwide. This allowed drinkers to buy tasting sets locally and compare the coffee themselves.
The idea was simple but powerful: to taste the same coffee in four versions, one with caffeine and three decaffeinated using different methods, so that the decaffeination process itself remained the only meaningful variable.
The results were more nuanced than many expected.
The reference coffee for the project was a Colombian lot with a clean, sweet profile, light fruit notes, moderate acidity, and balanced sweetness. Its clarity and lack of excessive aromatic complexity made it a useful baseline for comparison.
The CO₂ process showed a relatively gentle impact on the coffee. Acidity and body felt slightly reduced, but the overall profile remained very close to the original. The cup stayed recognizable, with fruit notes appearing a bit softer and more restrained. For many, this was surprising, as the influence of decaffeination turned out to be much less dramatic than expected.
The ethyl acetate (EA) version leaned slightly more toward fruit and sweetness. The cup showed pleasant sweetness, a smooth texture, and a clean, expressive aroma. Importantly, there was no sense that flavor had been stripped away by the removal of caffeine. The profile remained coherent and balanced.
The Swiss Water version felt fuller and rounder in the body. The texture seemed slightly heavier than in the original coffee, and the aroma was calmer, but the flavor remained rich. Overall, the cup came across as more substantial, with an emphasis on body and depth.
What this tells us about decaf
Overall, the project suggested something important: decaffeination tends to shift the balance rather than erase flavor. A good coffee can remain expressive even without caffeine; it simply presents itself a little differently in the cup.
Understanding how coffee is decaffeinated helps explain why different decaf coffees can taste so different. The processing method matters, but it’s only part of the story. The quality of the green coffee, the roasting approach, and the overall handling of the lot all play a major role in the final cup.
If there’s one clear takeaway from both research and tasting comparisons, it’s this: good decaf starts with good coffee.
Modern decaffeination doesn’t automatically mean dull or lifeless flavor. When done carefully, it tends to rebalance the cup rather than erase its character. The best decaf still reflects the origin, roast, and structure, just in a slightly different way.
Choosing decaf, in the end, isn’t about avoiding caffeine. It’s about choosing good coffee.